Scientists discover hidden aluminum sites that supercharge zeolite catalysis—this recent breakthrough is transforming our understanding of how zeolites work and how they can be optimized for cleaner energy, greener manufacturing, and more efficient chemical production. Whether you’re a student, a curious reader, or a professional in the field, this comprehensive guide will explain the science, the impact, and the practical opportunities of this discovery in a way that’s both approachable and authoritative.
Scientists Discover Hidden Aluminum Sites That Supercharge Zeolite Catalysis
| Aspect | Details |
|---|---|
| Discovery | Hidden aluminum sites in zeolites dramatically boost catalytic activity |
| Key Data | Over 60% increase in Brønsted acid sites after water activation in USY zeolite |
| Techniques Used | Advanced solid-state NMR, resonant X-ray diffraction, atom probe tomography |
| Applications | Oil refining, petrochemicals, green energy, environmental catalysis |
| Professional Impact | Enables rational design of more efficient, longer-lasting catalysts |
| Official Resource | Journal of the American Chemical Society |
The discovery of hidden aluminum sites that supercharge zeolite catalysis represents a major leap forward in both science and industry. By finally understanding where these powerful sites are and how to activate them, we can design better catalysts for a cleaner, more efficient, and more sustainable world. This breakthrough is already inspiring new research, new products, and new solutions to some of our biggest environmental and industrial challenges.
What Are Zeolites and Why Are They So Important?
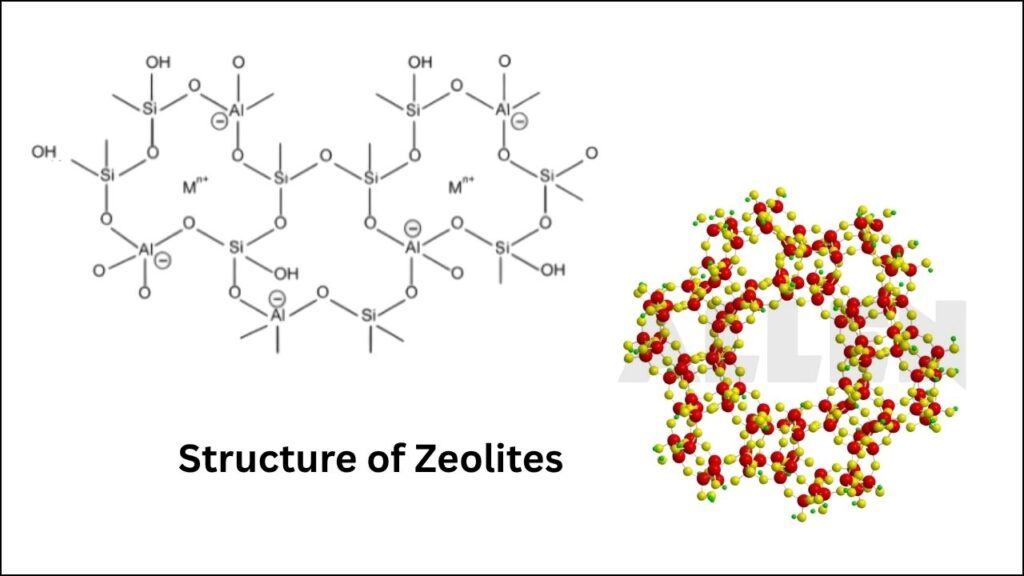
Zeolites are remarkable, sponge-like minerals made mostly of silicon, aluminum, and oxygen. Picture them as tiny honeycombs, filled with millions of microscopic holes and tunnels. These pores are just the right size to trap certain molecules while letting others pass—making zeolites nature’s perfect molecular sieves.
Everyday Uses of Zeolites
- Oil refining: Breaking down crude oil into gasoline, diesel, and jet fuel.
- Chemical manufacturing: Producing plastics, fertilizers, and detergents.
- Environmental cleanup: Removing pollutants from water and air.
- Plastic recycling: Converting waste plastics into valuable chemicals.
- Clean energy: Enabling sustainable fuel production, such as converting CO₂ into hydrocarbons.
Zeolites are used in over 40% of all acid-catalyzed industrial reactions, thanks to their unique structure and stability.
The Secret Power: Aluminum in Zeolites
Why Aluminum Atoms Matter
Inside a zeolite’s crystal structure, most atoms are silicon and oxygen. But when a silicon atom is replaced by an aluminum atom, it creates a negative charge. This negative charge is balanced by a proton (a hydrogen ion), forming a Brønsted acid site—the true engine of zeolite catalysis.
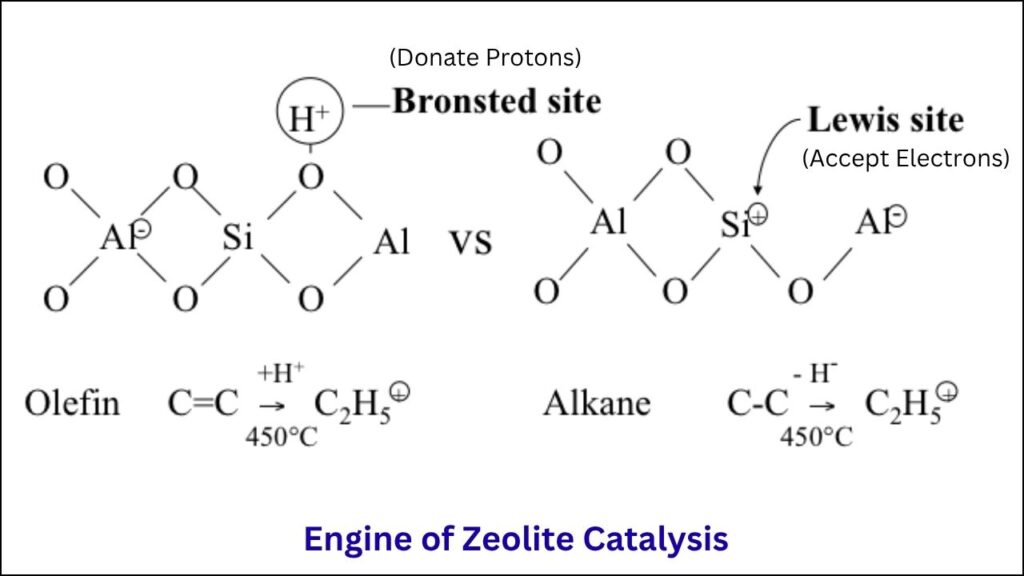
These acid sites are where the most important chemical reactions happen. They help break apart large molecules, join smaller ones together, and do so with incredible speed and efficiency. The number, location, and type of these aluminum-based acid sites determine how well a zeolite performs.
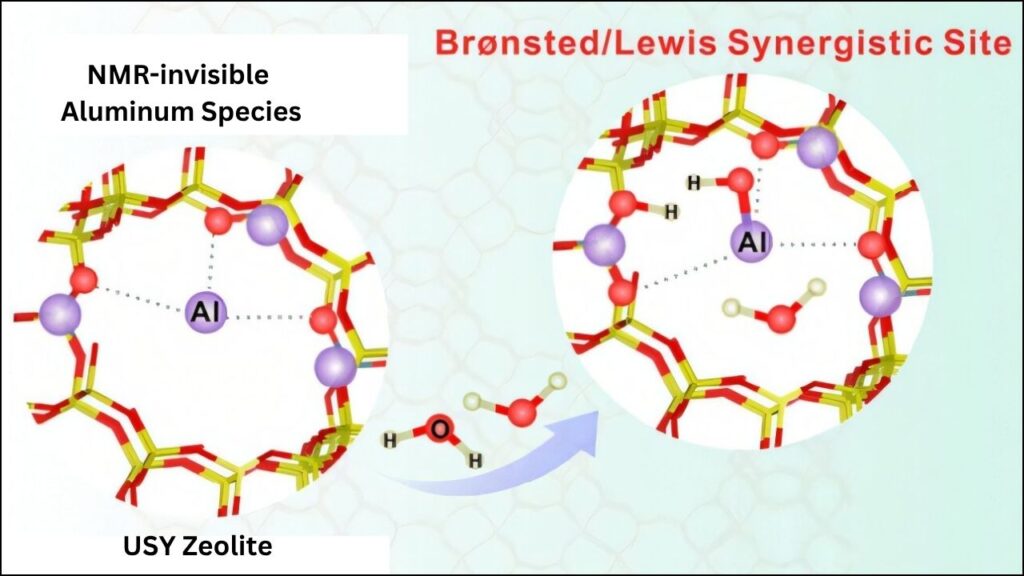
The Challenge: Finding Hidden Aluminum
For decades, scientists knew that not all aluminum atoms in zeolites were equally active. Some were easy to find and study, while others were “hidden”—invisible to traditional tools. This made it difficult to explain why some zeolites were much better catalysts than others, even when they seemed almost identical.
The Breakthrough: Discovering Hidden Aluminum Sites
New Tools, New Insights
With the development of advanced scientific techniques, researchers have finally been able to “see” these elusive aluminum atoms:
- Resonant X-ray diffraction: By tuning X-rays to the exact energy that aluminum absorbs, scientists can highlight aluminum atoms against the backdrop of silicon.
- Solid-state NMR (Nuclear Magnetic Resonance): This technique uses powerful magnets to probe the local environment of atoms, revealing “NMR-invisible” aluminum sites that were previously missed.
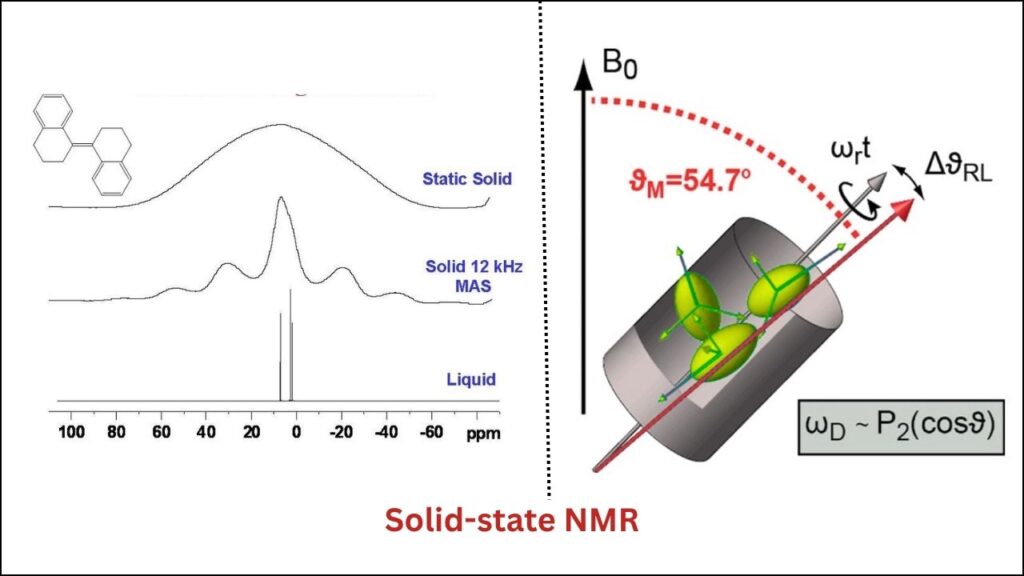
- Atom probe tomography: A 3D mapping tool that shows the exact position of atoms inside the zeolite, even at the nanoscale.
What Did Scientists Discover?
- Water-Activated Aluminum Sites: In ultra-stable Y (USY) zeolite, water molecules can “wake up” previously invisible aluminum sites. When water interacts with these sites, the number of Brønsted acid sites increases by more than 60%. This means the catalyst becomes much more powerful and efficient.
- Paired and Clustered Aluminum Sites: Some zeolites contain paired aluminum atoms or clusters near the surface. These create unique acid sites that are especially effective for certain reactions, such as converting methanol to gasoline or breaking down heavy oils.
- Extra-Framework Aluminum: In some cases, aluminum atoms are removed from their original positions and form new, highly active sites outside the main zeolite framework. These extra-framework sites can dramatically boost catalytic activity, especially for difficult reactions like hydrocarbon cracking.
Why Does This Matter? Real-World Impact
Supercharged Catalysts for Industry
With this new understanding, scientists and engineers can now design zeolites that are:
- More active: Speeding up reactions and increasing yields.
- More selective: Producing more of the desired product with less waste.
- Longer-lasting: Resisting deactivation and lasting through more cycles.
Example: In oil refineries, zeolites are used to crack heavy oil into gasoline. By tuning the aluminum sites, refineries can get more fuel from the same amount of crude oil, saving money and reducing emissions.
Greener Chemistry and Clean Energy
Optimized zeolites can make chemical manufacturing safer and more sustainable. For example:
- CO₂ hydrogenation: Zeolite-based catalysts are being used to convert carbon dioxide into valuable fuels and chemicals, helping fight climate change.
- Plastic recycling: Special zeolites can break down waste plastics into reusable materials.
- Ammonia-assisted NOx reduction: In automotive and industrial emissions control, zeolites help remove harmful nitrogen oxides from exhaust gases.
Environmental Benefits
Better zeolites mean:
- Cleaner air and water: More effective removal of pollutants.
- Reduced waste: Higher efficiency means less raw material and energy are needed.
- Lower greenhouse gas emissions: Improved catalysts for green fuel production and carbon capture.
How Do Scientists Study and Optimize Aluminum Sites?
Step 1: Mapping the Aluminum
Using advanced imaging and spectroscopy, researchers map where aluminum atoms are located inside the zeolite, including those that were previously hidden.
Step 2: Testing Catalytic Performance
Scientists run chemical reactions using these zeolites and measure how fast and efficiently they work. They also test how the catalyst performs under different conditions (temperature, pressure, presence of water).
Step 3: Rational Catalyst Design
With this information, researchers can:
- Change synthesis methods to place aluminum atoms exactly where they’re most effective.
- Pair aluminum atoms to create super-active sites for specific reactions.
- Treat zeolites with steam or chemicals to move or reveal aluminum atoms, optimizing the catalyst for its intended use.
Step 4: Enhancing Stability and Lifetime
By understanding how aluminum sites change during use (such as during hydrothermal aging in exhaust treatment systems), scientists can design zeolites that resist deactivation and maintain performance over time.
Practical Advice for Professionals
If you work in catalysis, petrochemicals, green chemistry, or environmental engineering, here’s how you can leverage these discoveries:
- Collaborate with research institutions to access and test new zeolite formulations with optimized aluminum sites.
- Monitor catalyst aging: Regular analysis of aluminum distribution can help predict and prevent performance drops.
- Consider water’s role: In some processes, controlled moisture can activate hidden aluminum sites and boost catalyst efficiency.
- Explore bimetallic and doped zeolites: Adding secondary metals (like copper, cerium, or alkali metals) can further enhance catalytic activity and stability, as shown in recent studies on ammonia-assisted NOx reduction and plastic recycling.
Cobalt-Based Catalyst Offers an Effective Alternative to Precious Metals
Hydrogen Fuel at Half the Cost? New Catalyst from South Korea
Precisely Placed Platinum Atoms Boost Catalyst Efficiency and Speed
FAQs About Scientists Discover Hidden Aluminum Sites That Supercharge Zeolite Catalysis
What are zeolites used for?
Zeolites are used in oil refining, chemical manufacturing, pollution control, plastic recycling, and clean energy production.
Why is aluminum important in zeolites?
Aluminum creates acid sites that are the heart of zeolite catalysis, enabling them to speed up and control chemical reactions.
How did scientists find the hidden aluminum sites?
They used advanced tools like resonant X-ray diffraction, solid-state NMR, and atom probe tomography to map and characterize aluminum atoms inside zeolites.
Can these discoveries make fuels and chemicals cheaper?
Yes. By making catalysts more efficient and durable, industries can produce more product with less energy and fewer raw materials, reducing costs and environmental impact.
The Future of Zeolite Catalysis: Opportunities and Challenges
Next-Generation Zeolites
The discovery of hidden aluminum sites opens the door to a new era in catalyst design. Scientists are now working on:
- Nanosheet zeolites: Ultra-thin zeolites with shorter diffusion paths, improving efficiency and reducing deactivation.
- Bimetallic and multi-functional zeolites: Combining different metals and active sites to tackle complex reactions, such as turning CO₂ into multi-carbon fuels or breaking down stubborn pollutants.
- Machine learning and AI: Using data-driven approaches to predict the best aluminum configurations for specific reactions.
Challenges Ahead
Despite these advances, several challenges remain:
- Controlling aluminum placement: Achieving precise control over aluminum distribution during synthesis is still difficult.
- Understanding deactivation: Coke deposition and framework collapse can still deactivate zeolites over time.
- Scaling up: Translating lab-scale breakthroughs to industrial-scale production requires further research and investment.