Clean energy is the future, and one of the most exciting breakthroughs today is a tiny iron catalyst that could change how we produce and use sustainable energy worldwide. This tiny but powerful innovation promises to make hydrogen fuel production cheaper, faster, and more eco-friendly than ever before. In this article, we will explore what this catalyst is, why it matters, and how it could impact industries, professionals, and everyday people.

Hydrogen fuel is considered one of the cleanest energy sources since it emits only water when used. The challenge has long been producing hydrogen efficiently and affordably. This new iron-based catalyst changes the game by offering a low-cost, highly efficient solution that works in regular water environments and renewable energy systems. Whether you are a professional in energy, technology, or a curious learner, this article breaks down the topic in simple language with deep insights to help understand and apply this innovation.
This Tiny Iron Catalyst Could Revolutionize the Future of Clean Energy
| Feature | Details |
|---|---|
| Catalyst Material | Iron-doped polymer-based complex (poly-Fe5-PCz) developed by researchers in Japan |
| Efficiency | Up to 99% Faradaic efficiency for water oxidation reactions, meaning nearly all electricity converts to oxygen |
| Stability | Exceptional long-term stability under rigorous conditions, outperforming precious metal catalysts like ruthenium |
| Cost-effectiveness | Uses abundant iron and nickel instead of scarce, expensive metals like platinum |
| Potential Applications | Hydrogen fuel production, clean energy storage, fuel cells, portable electronics, and green chemical synthesis |
| Environmental Impact | Non-toxic, abundant, and eco-friendly catalyst enabling greener industrial processes |
| Source / Reference | Institute of Science Tokyo – Catalyst Study (Nature Communications) |
The tiny iron catalyst represents a pivotal development in clean energy technology. It brings together cost-effectiveness, efficiency, and environmental sustainability to revolutionize how we produce and store hydrogen fuel. By replacing expensive precious metals with abundant iron and nickel, this innovation has the potential to make clean hydrogen fuel affordable and accessible worldwide.
For professionals in energy, materials science, and environmental fields, this catalyst opens new frontiers for research and commercial opportunities. For everyone else, it promises a cleaner, safer world powered by sustainable energy solutions. By combining the ingenuity of natural processes with cutting-edge technology, this breakthrough marks a step closer to a future where clean energy is a reality for all.
What Is a Catalyst and Why Is It Important for Clean Energy?
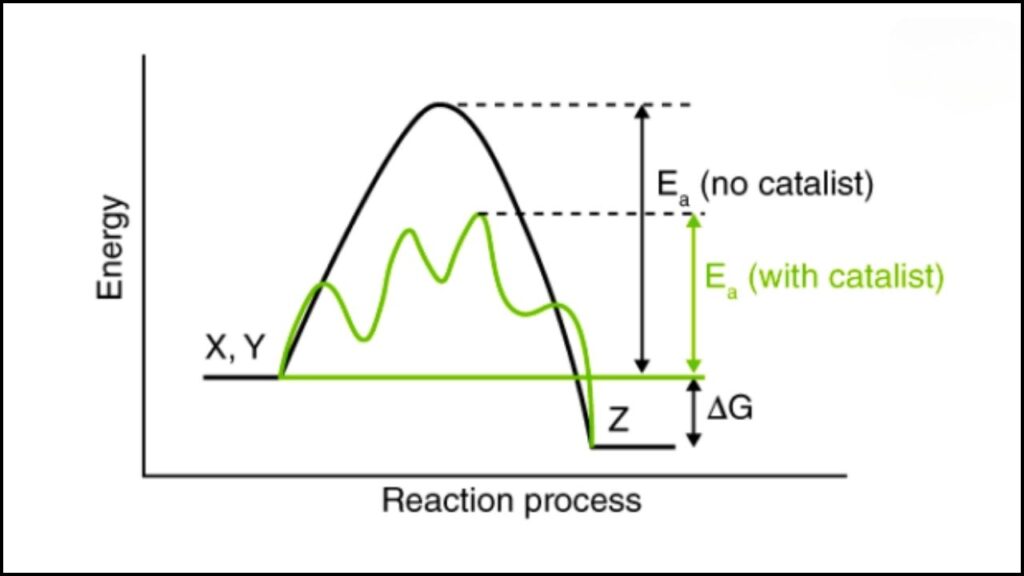
A catalyst is a special material that speeds up chemical reactions without being used up itself. Think of it as a helpful assistant that makes things happen faster and more efficiently. In clean energy, catalysts are crucial because they help convert water into hydrogen fuel and oxygen—a process called “water splitting.”
Traditional catalysts for this process often use rare, expensive metals like platinum or ruthenium, which limits how widely these technologies can be used due to high costs.
This is why scientists have been searching for affordable, effective alternatives using earth-abundant materials like iron and nickel. These metals are common, inexpensive, and much better for the environment.
The Breakthrough: Tiny Iron Catalyst
Recently, a team led by Professor Mio Kondo at the Institute of Science Tokyo developed a novel iron-based catalyst called poly-Fe5-PCz. This tiny catalyst cleverly combines iron atoms into a polymer material that mimics natural processes of oxygen production observed in plants during photosynthesis, known as water oxidation.
How Does It Work?
- The catalyst facilitates the oxygen evolution reaction (OER), a key step in splitting water into oxygen and hydrogen.
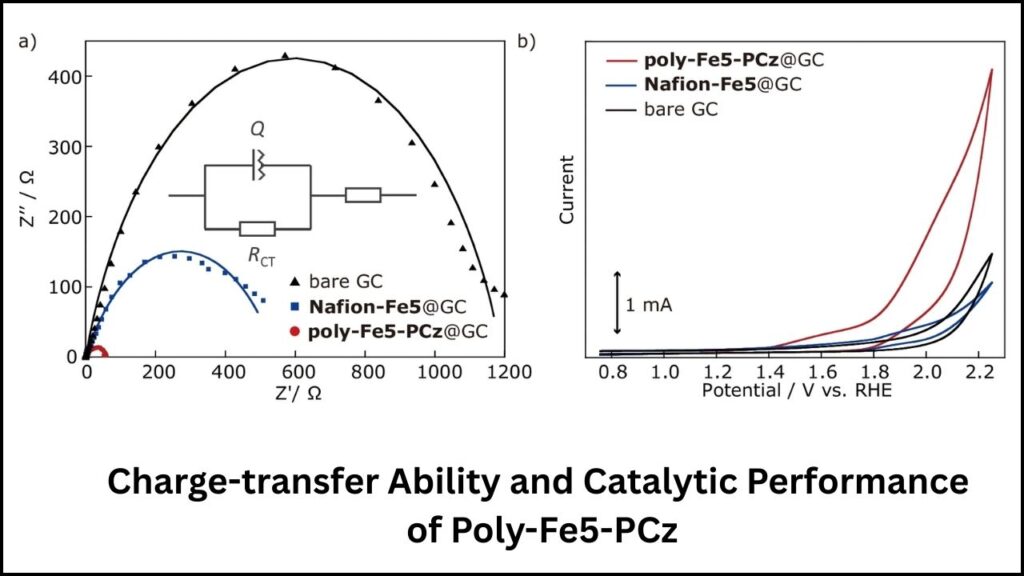
- It achieves up to 99% Faradaic efficiency, meaning almost every bit of electricity used is converted productively to oxygen, maximizing energy use.
- It remains stable and effective even under harsh conditions, ensuring it lasts longer in practical energy systems.
- Because it is made from iron and nickel, it is much cheaper than typical precious metal catalysts, making hydrogen fuel production more accessible and scalable.
Practical Impact and Applications
1. Hydrogen Fuel Production
Hydrogen is hailed as a future fuel for cars, trucks, planes, and even power plants because it produces only water when burned. Producing hydrogen fuel cleanly and affordably has been the missing piece. This new iron catalyst lowers the costs of electrochemical water splitting, making green hydrogen more competitive with fossil fuels.
2. Clean Energy Storage
Renewable energy like solar and wind sometimes produces more power than immediately needed. This extra energy can split water into hydrogen using the catalyst, storing energy chemically to be used later, balancing supply and demand efficiently.
3. Portable Electronics and Transportation
Hydrogen fuel cells powered by this catalyst could lead to longer-lasting batteries for electric vehicles and portable devices, reducing reliance on traditional batteries that use harmful materials.
4. Industrial Chemical Production
Besides energy, oxygen and hydrogen are prized in chemical industries. A cheap and durable catalyst means cleaner and cheaper production processes for chemicals like hydrogen peroxide and other green fuels.
Step-by-Step: How Scientists Developed the Iron Catalyst
- Synthesis of Catalyst Complex
Scientists synthesized a pentanuclear iron complex through organic chemical methods, ensuring precise molecular design for optimal activity. - Polymerization
Through electrochemical polymerization, the iron complex was transformed into a polymer coating on electrodes, creating a large number of active sites. - Testing for Efficiency and Stability
Using sophisticated tests like gas chromatography and electrochemical impedance spectroscopy, researchers confirmed the catalyst’s exceptional performance and durability. - Real-World Adaptation
Its performance under water and oxygen-rich environments showed it could directly replace or improve existing catalyst systems based on precious metals.
Why Is This Catalyst a Green Energy Gamechanger?
- Abundant and Non-toxic: Iron is the fourth most common element on earth, making it highly sustainable compared to precious metals.
- Lower Energy Use: High catalytic efficiency means less energy wasted.
- Durable: Long life reduces waste and replacement costs.
- Scalable: Easier to produce in large quantities at low cost.
- Multifunctional: Useful for various catalytic processes, enabling flexible applications.
New MOF Catalyst Developed for Efficient C–O Cross Coupling Reactions
FAQs About This Tiny Iron Catalyst Could Revolutionize the Future of Clean Energy
Q1: How does this catalyst compare to platinum or ruthenium?
A: It offers comparable or better efficiency and stability at a fraction of the cost thanks to the use of abundant iron and nickel instead of rare metals.
Q2: Where can this catalyst be used?
A: In hydrogen fuel cells, renewable energy storage, clean chemical manufacturing, and other electrocatalytic processes.
Q3: Is this catalyst ready for commercial use?
A: Research is progressing rapidly, with initial tests showing great promise. Scaling up production and integration into industrial systems are next steps.
Q4: Does it have environmental risks?
A: No. Iron and nickel are non-toxic and widely available, making this catalyst eco-friendly and safe for large-scale deployment.
Q5: How does this impact the future of clean energy jobs?
A: New technologies like this will create jobs in clean energy manufacturing, research, installation, and maintenance, supporting a green economy with sustainable growth.