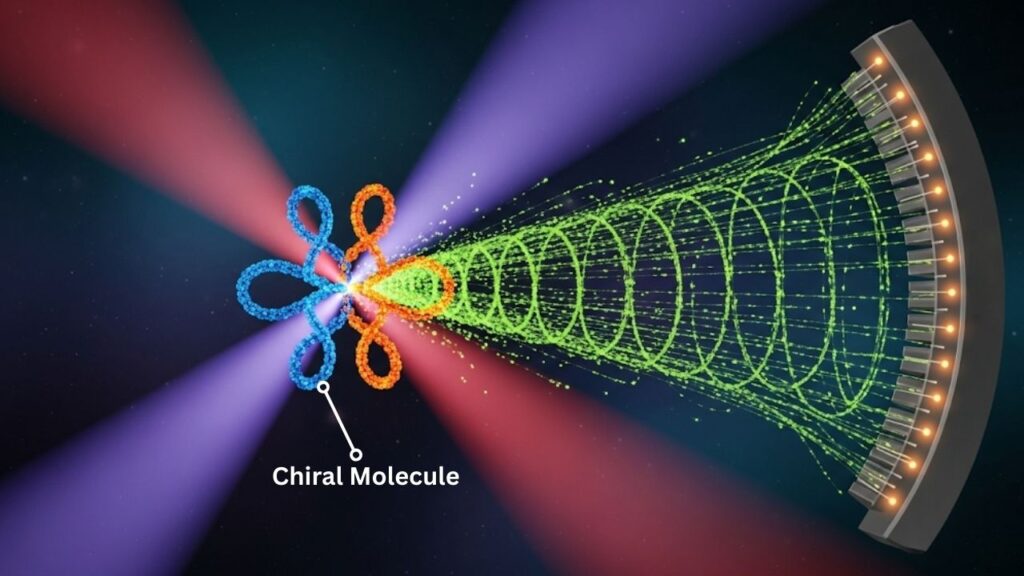
In a groundbreaking advancement announced in 2025, scientists have achieved attosecond control over chiral photoionization, marking a true quantum leap in the study of ultrafast electron dynamics within chiral molecules. This thrilling development enables researchers to observe and manipulate electron behavior in molecules that possess “handedness” (chirality) on their natural, ultrafast time scale—the attosecond (one quintillionth of a second). Such a breakthrough opens the door to more profound understanding and innovative control of molecular processes critical in fields like chemistry, pharmacology, and materials science.
If this sounds complicated, don’t worry! This article will break down this cutting-edge science in a simple, friendly way easily grasped by anyone, including a curious 10-year-old. At the same time, it will provide significant insights and practical information for professionals looking to stay ahead in their fields.
What Is Attosecond Control of Chiral Photoionization?
Chiral molecules are molecules that exist in two forms that are mirror images of each other, similar to left and right hands. This “handedness” matters hugely in biology and chemistry because two mirror-image molecules can behave very differently, for example, one could be therapeutic while the other harmful.
Photoionization is a process where light ejects electrons from a molecule, essentially knocking electrons out to see how the molecule responds. By controlling the light with attosecond precision—meaning billionths of billionths of a second—scientists can study exactly how these electrons move and interact in real time. When this is done to chiral molecules, it reveals how their “handedness” influences electron behavior.
Attosecond pulses of light, which last only a few hundred attoseconds, provide the precision necessary to see electron dynamics that were previously too fast to observe or control. These pulses can be circularly polarized (twisting in a circular motion), which interacts particularly well with chiral molecules.
Scientists Achieve Attosecond Control of Chiral Photoionization
| Aspect | Details |
|---|---|
| Breakthrough | Attosecond coherent control of chiral photoionization dynamics achieved |
| Time Scale | Electron dynamics controlled within ~100 attoseconds (1 attosecond = 10⁻¹⁸ seconds) |
| Method | Use of circularly polarized attosecond XUV pulses combined with near-infrared light |
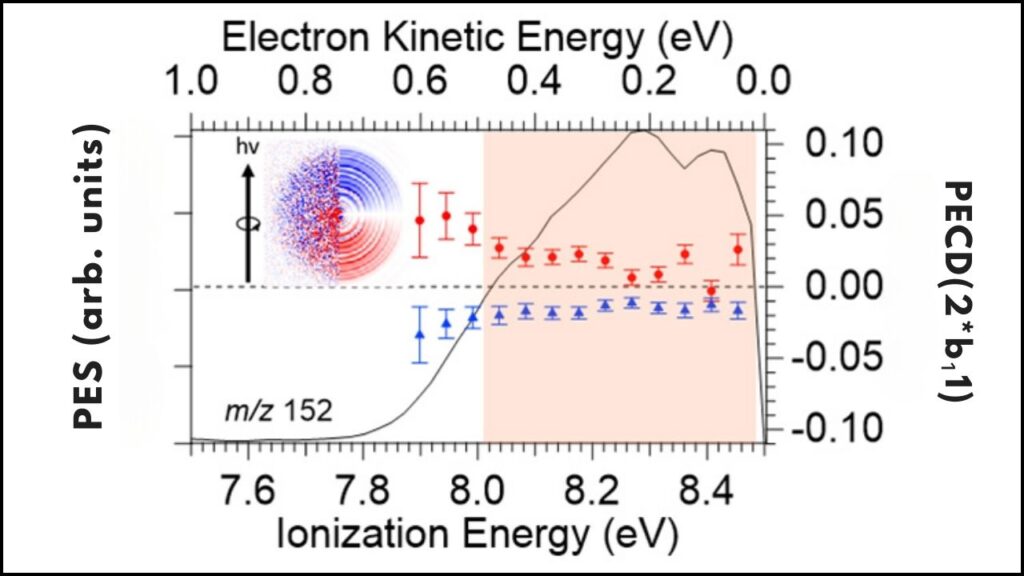
| Impact on PECD (Photoelectron Circular Dichroism) | Nearly doubled PECD signals, with ability to invert signal sign |
| Photoionization Delays | Measurable chiral-dependent delays up to 240 attoseconds in electron emission angles |
| Applications | New understanding of chiral molecules, pharmaceutical development, spintronics, molecular physics |
| Official Paper | Read Original Research on arXiv |
The achievement of attosecond control over chiral photoionization represents a monumental leap in science, providing unprecedented power to probe and manipulate electron dynamics in chiral molecules. This innovation enhances our fundamental understanding of molecular processes and opens up exciting new pathways in chemistry, photonics, and quantum technologies. Both researchers and practitioners can look forward to future applications that leverage this precision, ultimately impacting fields from drug development to spintronics.
Why Is This Important?
For decades, chiral molecules have been vital in fields like drug design, where the difference between left- and right-handed versions of molecules can mean the difference between a life-saving medicine and a dangerous compound. By understanding how electrons behave in these molecules on their natural time scale (attoseconds), scientists can gain unparalleled insights into chemical reactions, molecular electronics, and even the fundamental causes behind phenomena like the chirality-induced spin selectivity (CISS) effect.
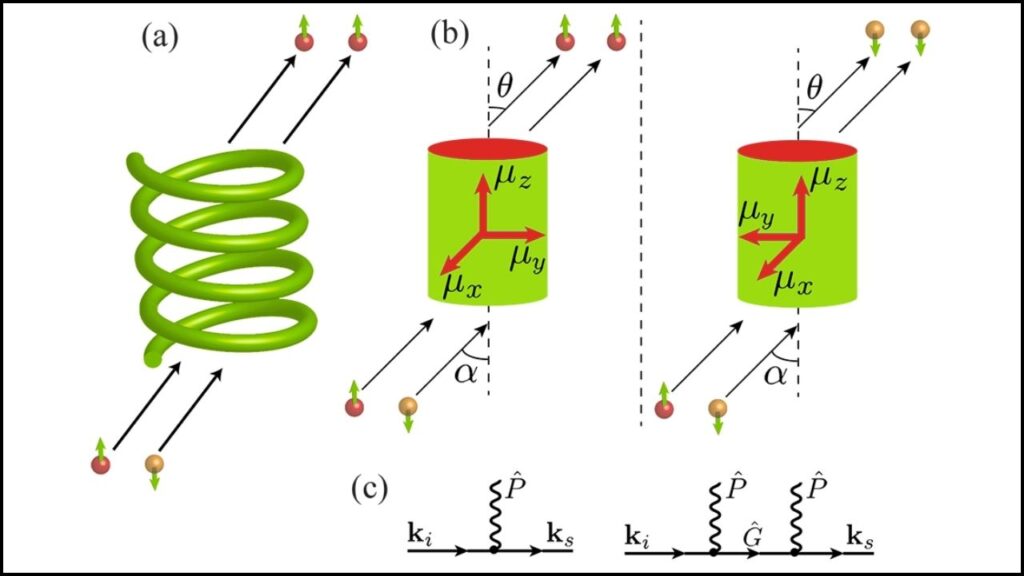
The ability to control these electron dynamics also means that new types of targeted light-based treatments or ultra-precise molecular sensors could be developed in the future, offering innovations across medicine and nanotechnology.
How Was This Breakthrough Achieved?
The success in controlling chiral photoionization comes from:
1. Generating Circularly Polarized Attosecond Pulses
Traditionally, it was difficult to create attosecond pulses of light that were circularly polarized, as this polarization is necessary for interacting with chiral molecules in a meaningful way. Researchers recently overcame significant technical challenges, producing these specialized pulses with unprecedented stability and control.
2. Combining Attosecond XUV Pulses and Near-Infrared (IR) Light
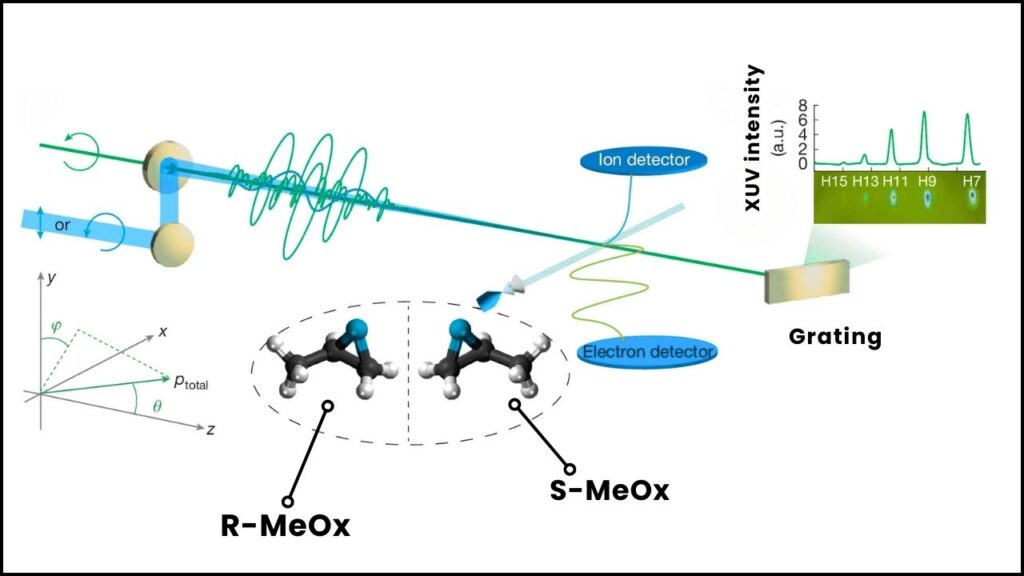
The researchers combined extreme ultraviolet (XUV) attosecond pulses with near-infrared pulses that rotate similarly (co-rotating) to enhance the photoelectron circular dichroism (PECD) effect. This combination nearly doubled PECD signals and even allowed scientists to invert its sign, showing powerful coherent control over the photoionization process.
3. High-Resolution Electron Detection
Using momentum-vector-resolved electron-ion-coincidence spectroscopy, the team captured detailed data on how electrons were ejected at different angles and energies. This method allowed for precise measurements of photoionization time delays—how long electrons took to escape—revealing new chiral-dependent asymmetries.
4. Sophisticated Data Analysis and Theoretical Modeling
The team applied advanced quantum mechanical models to simulate and explain the observed electron dynamics, confirming that these chiral signatures were authentic and measurable.
Practical Insights for Professionals
For scientists, engineers, and technology developers, this advancement means:
- Enhanced Analytical Tools: Attosecond chiroptical spectroscopy allows unprecedented access to electronic motions in chiral molecules, which can improve drug discovery, materials design, and molecular electronics.
- Better Understanding of Spintronics: By directly measuring chiral electron delays and asymmetries, the underlying physics of spin-selective transport (CISS), crucial for next-generation electronics, can be unraveled.
- Future Technologies: The precision control of electron motion can aid the development of ultrafast optical switches, quantum computers, and better sensors.
Step-by-Step Guide to Attosecond Chiral Photoionization Control
Step 1: Prepare the Chiral Molecule Sample
Choose the chiral molecules to study, ensuring they are available in pure left- or right-handed forms (enantiomers).
Step 2: Generate Attosecond Circularly Polarized Light
Use a high-harmonic generation setup, which involves shooting intense lasers at gases to produce attosecond XUV pulses with circular polarization.
Step 3: Apply Co-Rotating Near-Infrared Fields
Combine the attosecond XUV pulses with synchronized near-infrared light fields also circularly polarized in the same rotational direction to enhance control over the photoelectron emission.
Step 4: Measure Photoelectron Emission
Use a momentum-resolved electron-ion coincidence spectrometer to capture the energy and angle of emitted electrons from the chiral molecules.
Step 5: Analyze Time Delays and PECD
Evaluate the time delays in electron emission and the photoelectron circular dichroism signals to understand the electron dynamics with attosecond precision.
Step 6: Model and Interpret Data
Use quantum mechanical simulations to explain the experimental results and predict how to modify light fields for desired control.
Attosecond ‘Clock’ Measures Electron Tunneling Time With Unmatched Precision
FAQs About Scientists Achieve Attosecond Control of Chiral Photoionization
Q1: What is an attosecond?
An attosecond is an unimaginably short fraction of a second—a billionth of a billionth of a second (1×10⁻¹⁸ seconds). It’s the time scale at which electrons move in atoms and molecules.
Q2: Why is chirality important?
Chirality determines how molecules interact in biological systems, pharmaceuticals, and materials. Two mirror-image molecules (enantiomers) can have entirely different effects.
Q3: What is photoionization?
Photoionization is when light knocks electrons out of atoms or molecules, allowing scientists to study their behavior and structure.
Q4: How does this research impact everyday life?
By understanding and controlling these ultrafast electron processes, we can develop better medicines, safer chemicals, and advanced technology like quantum computers.
Q5: Where can I learn more about this technology?
Visit high-authority resources like arXiv.org for detailed scientific papers, or scientific news portals such as Science.org for digestible updates.