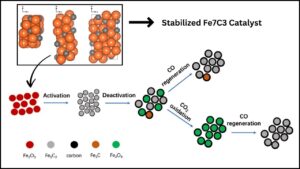
Carbon dioxide (CO2) hydrogenation to olefins is a promising sustainable technology that can turn greenhouse gases into valuable chemicals and fuels. Among various catalysts researched, the stabilized Fe7C3 catalyst with potassium (K) and magnesium (Mg) promotion has recently emerged as a breakthrough, significantly advancing CO2 conversion efficiency and selectivity towards high-value olefins. This article explores the science of this catalyst, its importance for the environment and industry, and practical insights on how it can shape the future of sustainable chemistry.
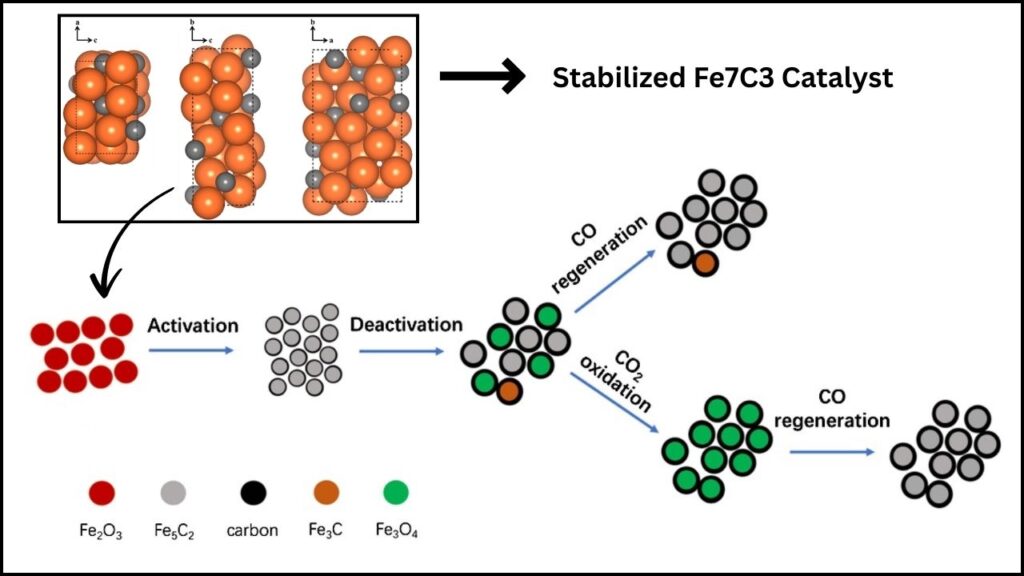
Table of Contents
Stabilized Fe7C3 Catalyst
| Feature | Details |
|---|---|
| Catalyst | Stabilized Fe7C3 catalyst with K-Mg dual promotion |
| Reaction | CO2 hydrogenation to light olefins |
| CO2 Conversion Rate | 41.5% at 340 °C and 2 MPa |
| Olefin Selectivity | 67.1% |
| Catalyst Stability | Over 1000 hours of stable operation |
| Key Benefits | Enhanced phase stability, olefin selectivity, water oxidation suppression |
| Source | Nature Communications, ACS Catalysis (2025) |
| Official Reference | Nature Communications Article |
The stabilized Fe7C3 catalyst with K-Mg dual promotion represents a game-changing advancement in the field of CO2 hydrogenation, enabling efficient and durable conversion of CO2 into high-value olefins. This innovation not only promises economic benefits but also aligns with global sustainability goals to reduce carbon emissions. Through stable phase control and smart chemical promotion, this catalyst stands at the forefront of turning waste CO2 into valuable building blocks for the chemical industry — a testament to how science meets sustainability in practical ways.
Understanding CO2 Hydrogenation and Olefins
CO2 hydrogenation is a chemical process where CO2 reacts with hydrogen (H2) to produce hydrocarbons like olefins—unsaturated hydrocarbons widely used as building blocks in plastics, detergents, and synthetic fibers.
Converting CO2, a major greenhouse gas, into these valuable products:
- Mitigates climate change by reducing atmospheric CO2.
- Adds economic value by creating useful chemicals from waste gas.
- Provides new sustainable pathways for industrial chemicals.
However, CO2 molecules are chemically stable, making this reaction challenging. Catalysts accelerate the process by lowering energy barriers and directing product formation. Scientists have explored iron-based catalysts, such as iron carbides, for their efficiency and abundance.
What Makes the Fe7C3 Catalyst Special?
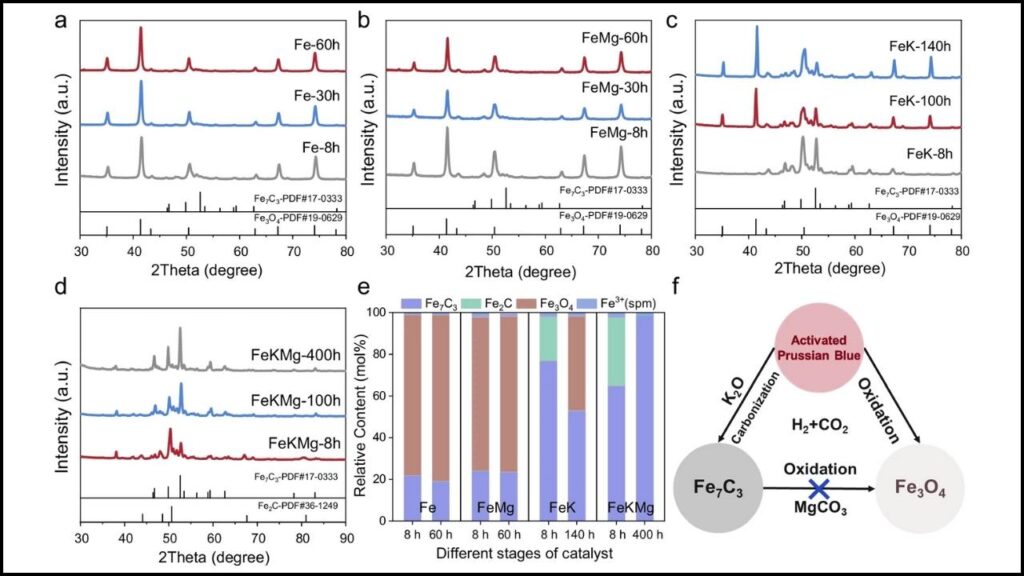
Iron carbide, notably the Fe7C3 phase, is known to have excellent catalytic activity for CO2 hydrogenation. Still, it faces two major issues:
- Phase Stability: Fe7C3 tends to degrade or transform under reaction conditions, reducing its catalytic efficiency.
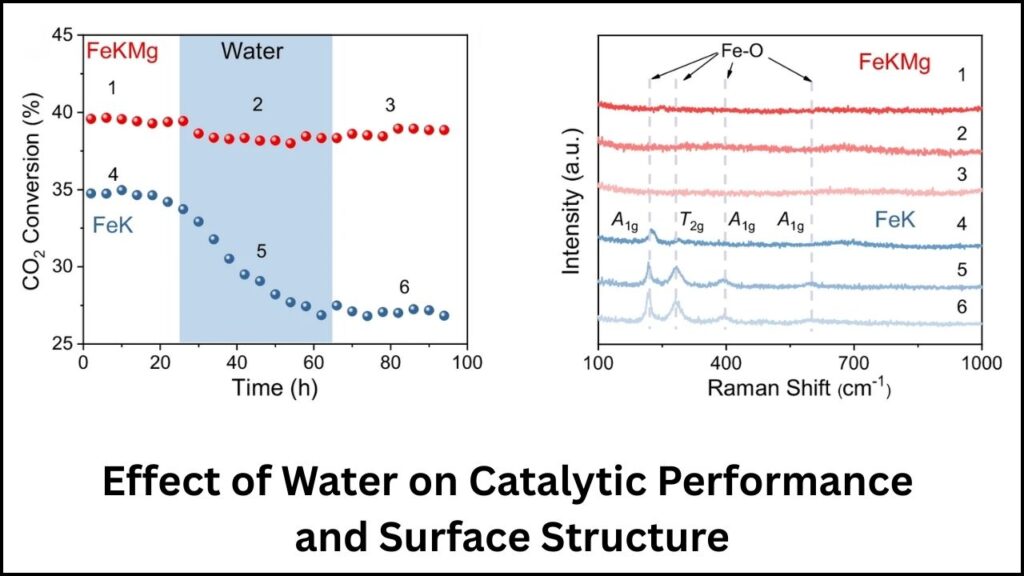
- Water Oxidation: Water formed during the reaction can oxidize the catalyst, damaging its structure.
The stabilized Fe7C3 catalyst solves these problems by incorporating two promoters:
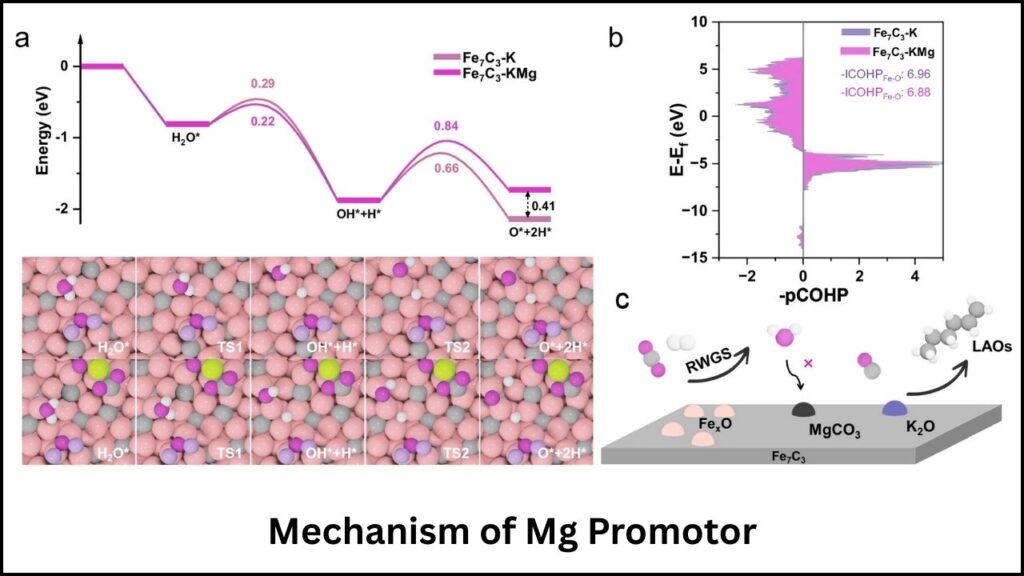
- Potassium (K): Accelerates the carbonization process and boosts olefin selectivity by optimizing reaction pathways.
- Magnesium (Mg): Prevents water-induced oxidation, preserving the catalyst’s crystalline structure during long-term use.
Researchers synthesized this catalyst from Prussian blue analogues, creating a near pure Fe7C3 phase with excellent stability.
Impressive Performance Metrics
Under optimized conditions (340 °C temperature, 2 MPa pressure, and H2/CO2 ratio of 3):
- CO2 conversion reaches 41.5% — an indicator of how much CO2 is turned into products.
- Olefin selectivity hits 67.1%, meaning two-thirds of the products are valuable olefins rather than byproducts.
- Stability is proven over 1000 hours without significant loss of activity, outperforming many existing catalysts.
These results mark a significant advance for industrial CO2 hydrogenation, making the process more efficient, economical, and durable.
Practical Insights: What This Means for Industry and Environment
Sustainable Chemical Production
Replacing fossil-feedstock with CO2-derived olefins can reduce carbon footprints across industries such as plastics, fuel, and chemicals.
Economic Benefits
The use of iron, a cheap and abundant metal, along with promoters like potassium and magnesium, creates a cost-effective catalytic system, promising scalable applications.
Long Catalyst Life
The enhanced stability reduces downtime and replacement costs in industrial reactors, improving process economics.
Step-by-Step Guide to CO2 Hydrogenation Using Fe7C3 Catalyst
- Catalyst Preparation:
- Synthesize Fe7C3 phase from Prussian blue analogues.
- Introduce potassium and magnesium as promoters to enhance performance and stability.
- Reaction Setup:
- Feed CO2 and H2 gases with a molar ratio of about 1:3.
- Maintain reaction pressure around 2 MPa and temperature near 340 °C.
- Catalytic Reaction:
- The Fe7C3 catalyst facilitates CO2 hydrogenation.
- Potassium directs pathway to form olefins; magnesium protects catalyst structure.
- Product Collection:
- High yield of light olefins such as ethylene and propylene.
- Byproducts like methane and CO are minimized.
- Catalyst Regeneration & Monitoring:
- Monitor catalyst performance to sustain activity over prolonged durations.
- Utilize promoters to prevent oxidation and phase degradation.
This Tiny Iron Catalyst Could Revolutionize the Future of Clean Energy
New MOF Catalyst Developed for Efficient C–O Cross Coupling Reactions
FAQs About Stabilized Fe7C3 Catalyst
Q1: Why is converting CO2 to olefins important?
A1: It helps reduce greenhouse gases and creates valuable chemicals for various industries, contributing to both climate action and economic gain.
Q2: What exactly are olefins?
A2: Olefins (like ethylene and propylene) are unsaturated hydrocarbons used to make plastics, adhesives, and chemicals essential in everyday life.
Q3: How does potassium improve the catalyst?
A3: Potassium accelerates the carbonization process in the catalyst and increases the selectivity toward olefins, enhancing the yield of valuable products.
Q4: What role does magnesium play in the catalyst?
A4: Magnesium acts as a stabilizer by suppressing water-induced oxidation of the Fe7C3 catalyst, extending its lifespan.
Q5: How long does this catalyst last?
A5: The stabilized Fe7C3 catalyst maintains exceptional performance for more than 1000 hours under reaction conditions.
Q6: Can this catalyst be used at a large scale industrially?
A6: Due to its stability, high efficiency, and cost-effectiveness, it shows great promise for scale-up and industrial application.