The ultrafast X-ray laser captures motion of a single electron for the first time, marking a groundbreaking milestone in the world of science. This breakthrough allows scientists to observe, in real time, the incredibly fast and tiny movements of an electron during chemical reactions. But what does this mean for science, technology, and even everyday life? Let’s explore this exciting discovery in a way that everyone—from curious kids to seasoned professionals—can understand and appreciate.
What Is an Ultrafast X-Ray Laser?
Imagine a super-powered flashlight that can take pictures not just of objects, but of events happening so fast they’re almost impossible to see, such as electrons moving inside atoms. That’s what an ultrafast X-ray laser does. Using extremely short, bright bursts of X-ray light, these lasers capture snapshots of the tiniest particles at unprecedented speeds, measured in femtoseconds (a millionth of a billionth of a second). This technology is far faster and more precise than any ordinary camera.
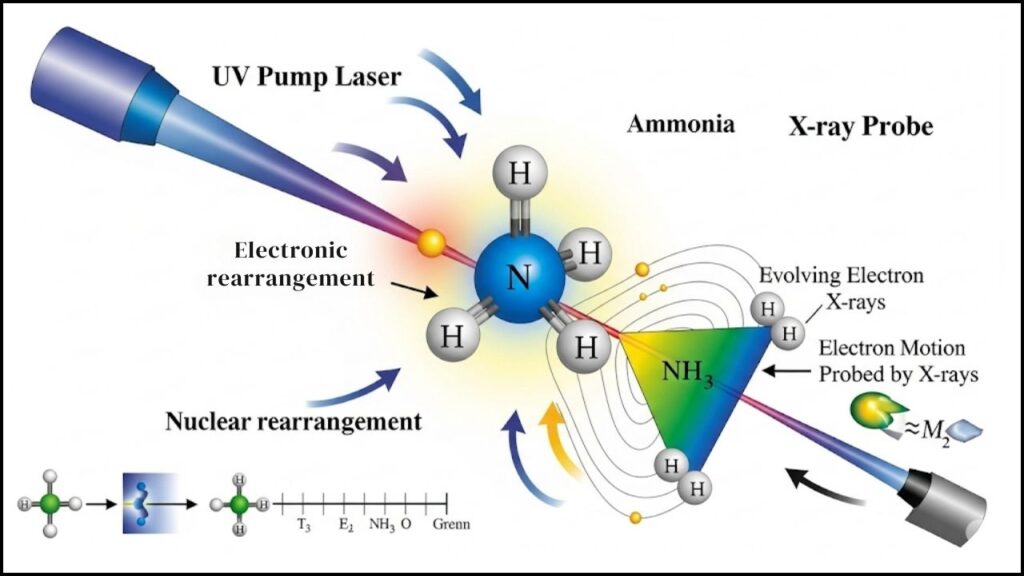
Recently, scientists at the SLAC National Accelerator Laboratory successfully used this laser to visualize the motion of a single valence electron—the electron responsible for chemical bonding—during a reaction involving ammonia molecules.
Ultrafast X-Ray Laser Captures Motion of a Single Electron for the First Time
| Aspect | Details |
|---|---|
| Technology Used | Ultrafast X-ray laser at SLAC’s Linac Coherent Light Source (LCLS) |
| Achievement | First real-time observation of a single valence electron in a chemical reaction |
| Time Resolution | About 500 femtoseconds (quadrillionths of a second) |
| Molecule Observed | Ammonia (NH₃) |
| Research Publication | Physical Review Letters, August 2025 |
| Lead Researchers | Ian Gabalski, Philip Bucksbaum, Nanna List |
| Practical Applications | Improved understanding of chemical reactions, advanced materials development, technology innovation |
| Official Website | SLAC National Accelerator Laboratory |
The ultrafast X-ray laser capturing the motion of a single electron represents a monumental advance in science. For the first time, researchers can watch an electron as it guides chemical reactions, opening the door to innovations across chemistry, materials science, energy, and technology. This breakthrough, made possible by cutting-edge lasers and computational analysis, ushers in a new era where observing and controlling the very building blocks of matter becomes achievable. It’s a leap forward that holds enormous promise, both for scientific understanding and practical applications that touch everyday life.
Why This Discovery Matters
Every chemical reaction involves electrons moving and interacting, but their rapid, unpredictable movements have made it almost impossible for scientists to watch them in action—until now. By capturing these electron motions:
- Scientists verify and refine chemical theories.
- Researchers can design better catalysts that speed up industrial reactions.
- The understanding gained aids in developing new materials and medicines.
- It opens up possibilities for controlling chemical reactions with precision, improving energy efficiency and sustainability.
How Did Scientists Capture an Electron’s Motion?
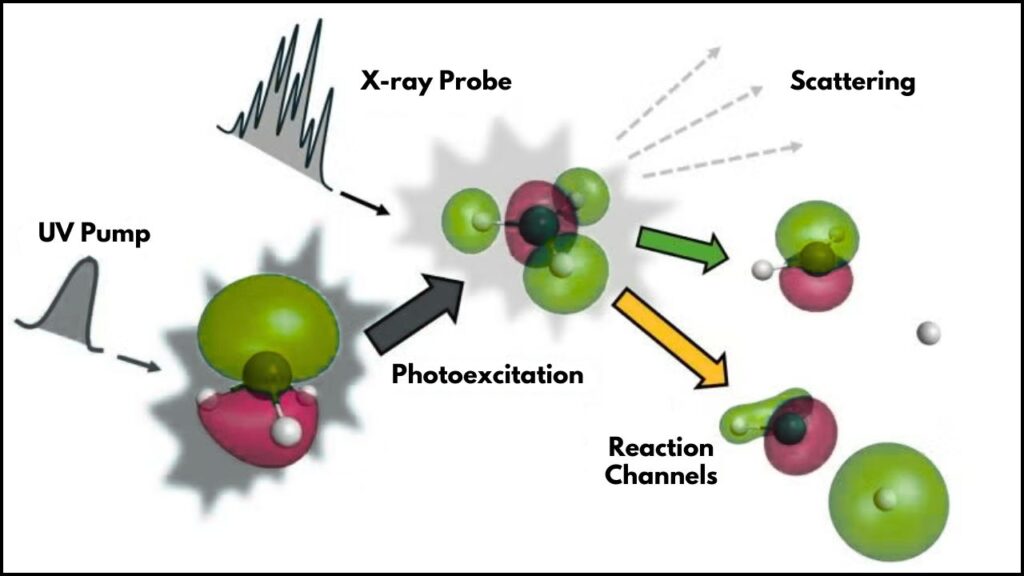
To understand how they did this, let’s break it down step by step:
1. Choosing the Right Molecule: Ammonia
Scientists selected the ammonia molecule (NH₃) because of its simplicity and well-known behavior. Ammonia is a common building block in chemistry, so observing it is like watching a classic example to learn from.
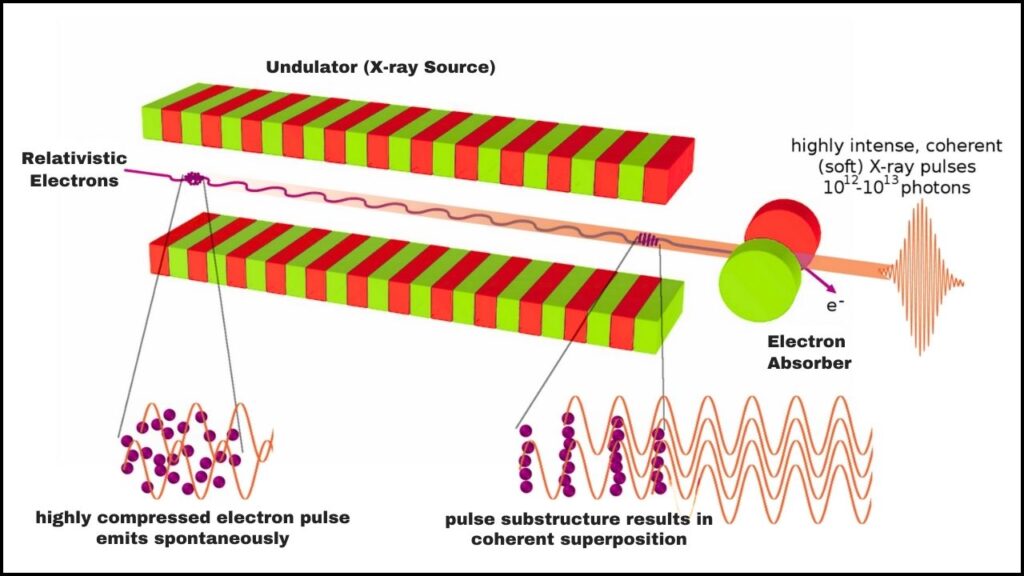
2. Using the Linac Coherent Light Source (LCLS)
LCLS, part of the SLAC laboratory, is one of the world’s most advanced X-ray free-electron lasers. It produces ultrashort X-ray pulses that act like an ultra-slow-motion camera allowing scientists to take rapid snapshots of electrons in action.
3. Timing the Reaction with Femtosecond Precision
Using precisely timed laser pulses, scientists kicked off a reaction by exciting the ammonia molecules. Then, at exactly the right moments, they captured the electron’s movement during the breaking of a hydrogen bond—a key part of the dissociation process.
4. Scattering and Data Collection
As the X-rays hit the ammonia molecules, electrons scatter the X-rays uniquely. Scientists collected thousands of patterns of this scattered light to map the exact path the electron took.
5. Advanced Simulations and Analysis
This huge amount of data was fed into sophisticated computer models and quantum theories to isolate and track the single valence electron’s behavior amid the noise of many core electrons.
Practical Implications for Science and Industry
Tracking electron motions in real time isn’t just a cool trick; it stands to transform many fields:
- Chemistry and Biochemistry: Understanding electron behavior could improve drug design by revealing how molecules interact at the most fundamental level.
- Energy Research: More efficient solar cells and batteries can be developed by controlling electron flow better.
- Materials Science: New materials with custom properties, such as superconductors or lightweight alloys, could emerge by controlling chemical reactions at the electron level.
- Nanotechnology: Precise control of electron dynamics could accelerate the development of nanoscale devices and quantum computers.
Breaking Down the Technology: What Makes It Possible?
- X-ray Free Electron Lasers (XFELs): Unlike traditional lasers, XFELs create ultra-bright, extremely short bursts of X-rays enabling atomic-scale resolution.
- Femtosecond Time Scale: Electrons zip around atoms in femtoseconds. Capturing that demands ultrafast lasers releasing pulses just a few femtoseconds apart.
- Valence vs. Core Electrons: Valence electrons determine chemical properties, but are harder to isolate than core electrons. This method specifically tracks valence electrons in real time.
- Data Analysis and AI: Huge volumes of data require sophisticated software to disentangle signals and reconstruct electron trajectories.
New Additive Method Transforms Perovskite Lasers — What This Means for Next-Gen Devices
Lasers Used To Push Crystal Vibrations To Their Quantum Ground State
New Timed Laser Method Lets Scientists Hit ‘Pause’ on Ultra-Fast Silicon Melting
FAQs About Ultrafast X-Ray Laser Captures Motion of a Single Electron for the First Time
What is a valence electron?
A valence electron is an electron located in the outermost shell of an atom. It plays a key role in chemical bonds and reactions.
How fast do electrons move?
Electrons move incredibly fast—on the timescale of femtoseconds (one quadrillionth of a second).
Why is it important to visualize electron motion?
Seeing electron motion allows scientists to understand how chemical reactions happen fundamentally, improving our ability to control and optimize these processes.
Can this technology be used outside labs?
Currently, ultrafast X-ray lasers require huge and expensive facilities like SLAC. However, technological advances could miniaturize this in the future.
How does this discovery impact everyday life?
Advances in chemistry, materials, and energy from this research can lead to better medicines, cleaner energy, and smarter technology, benefiting everyone.